 
Here is the notation of IUPAC nomenclature of elements Highest principle quantum number(n) or Principal Valence ShellĤ) Within a period, electron is filled in same valence shells.ĥ) Within a period, atomic size decreases due to increase in effective nuclear charge. The number of elements in the period is equal to the number of electrons which can be accommodated in the orbitals belonging to that electron shell of shells)ģ) The period corresponds to highest principle quantum number(n) of the elements in the period. 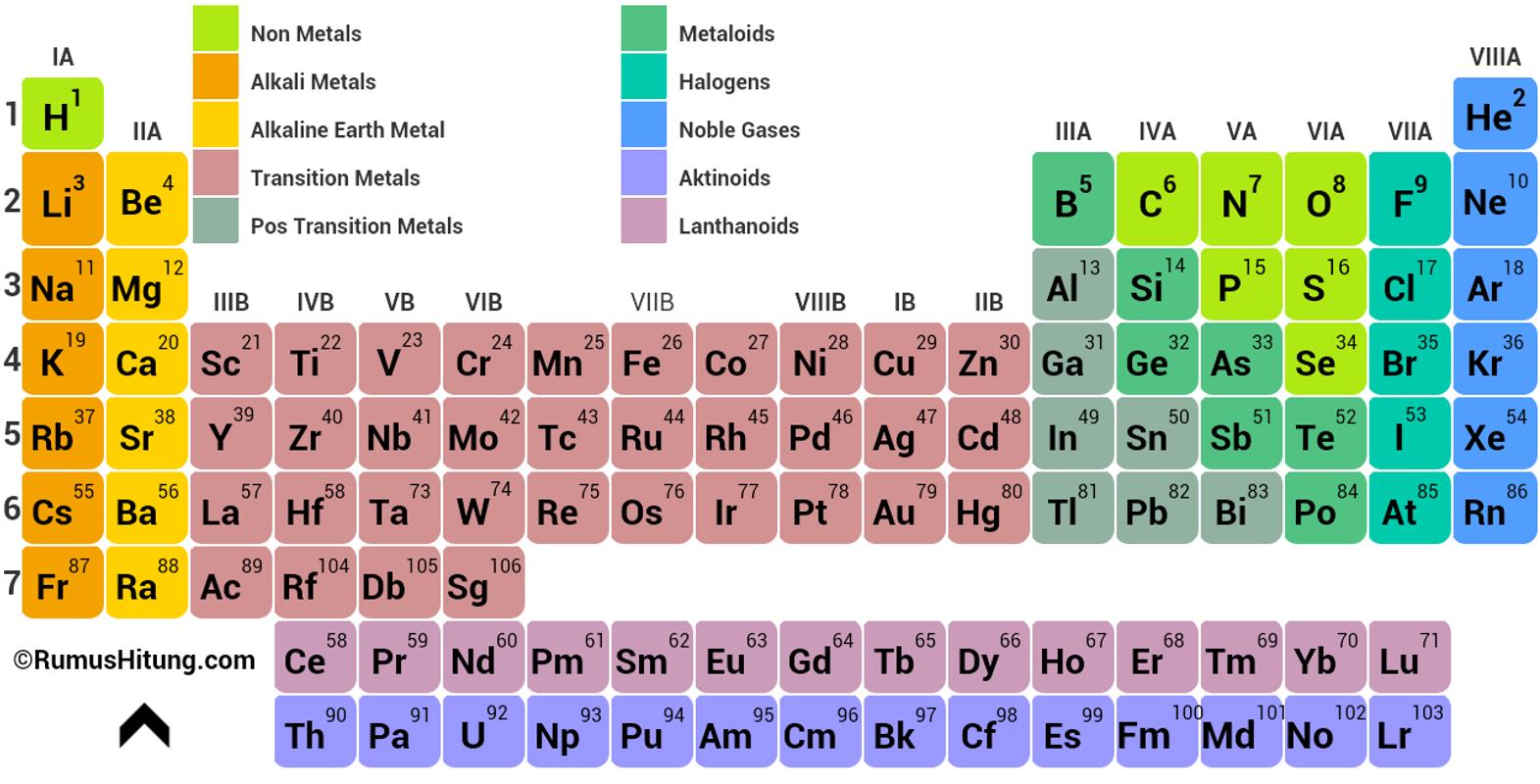
& atomic size increases on going down the group (due to increase in no. There are total 18 groups numbered 1 to 18.Īll elements in a group have same properties in general.Ītomic no. Elements have similar outer electronic configuration are arranged in same groups. There are altogether 7 periods in the Periodic tableĢ) Vertical columns are called groups. They were called series in Mendeleev Periodic Table. “The physical and chemical properties of elements are the periodic functions of their atomic numbers”Ī modern version which is called Long Form of the Periodic Table is most widely used and acceptedġ) Horizontal rows are called period. So, Mendeleev Periodic Table was modified and it is called now Modern Periodic Table which states that 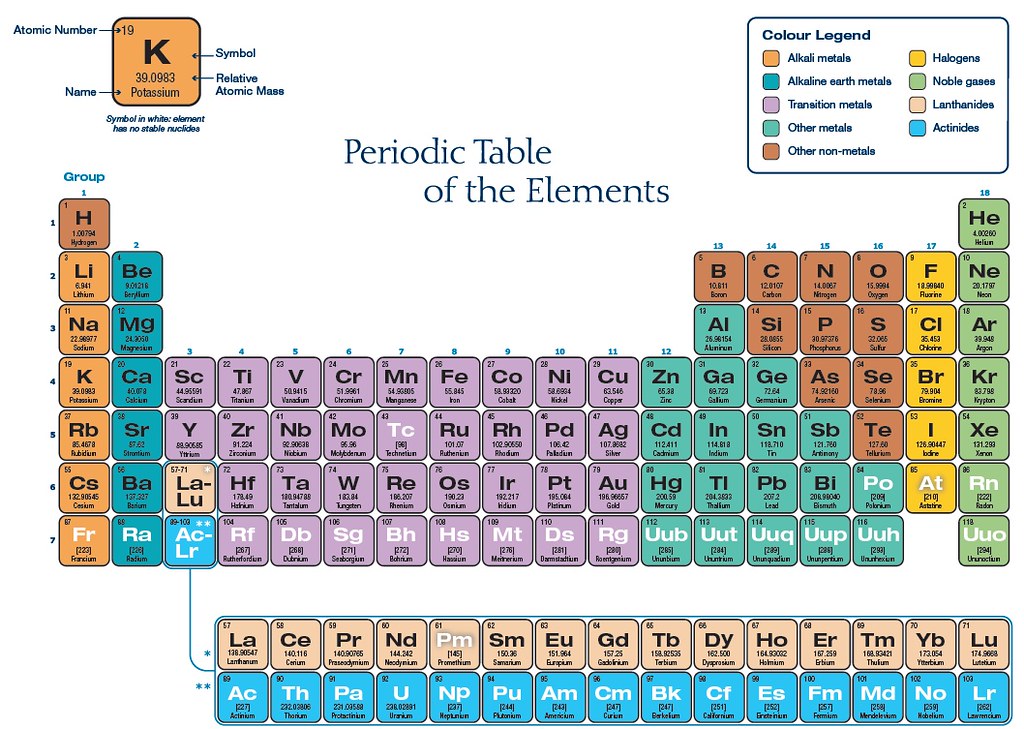
Therefore, the position of an element in the periodic table depends on its atomic number than its atomic mass. Moseley, the English physicist showed that atomic number is more fundamental property of an element than its atomic mass. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
